Meaning of resonance in chemistry8/13/2023 
Specific charges assigned to the required atom in a compound or molecule is called formal charge. Now the central oxygen atom is represented by a positive charge.Īgain the oxygen atom having no charge i.e neutral oxygen atom donates its electron pair to a positively charged central atom resulting in the neutral oxygen atom getting a negative charge. Hence, the oxygen atom at the centre donates its electrons to the positively charged oxygen atom. Lewis Dot Structure for O 3.Īccording to the given structure, (structure 1) one oxygen atom is positively charged and the other one is negatively charged. 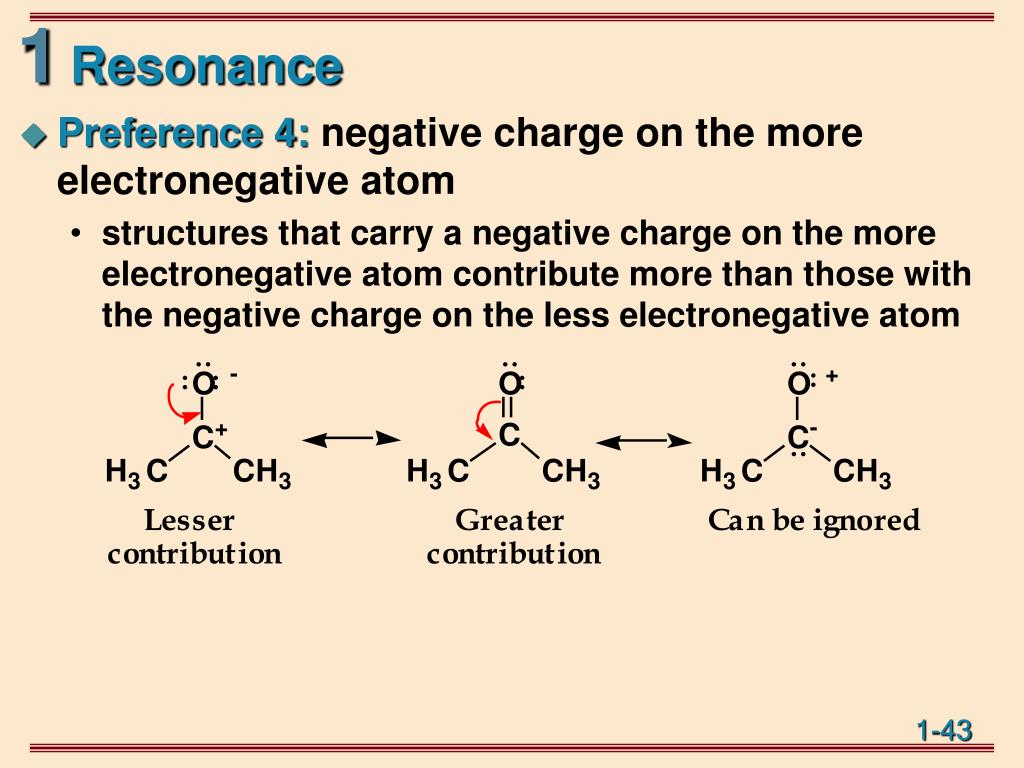
We know that oxygen is a bivalent atom and forms bonds with two other oxygen atoms in the ozone molecule. Now, let us have a look at the O 3 molecule or OZONE which is a V-shaped structure. The number of lone pairs of electrons must remain the same as the at of the original structure. The hybridization of the structure must not be changed. Only the position of electrons is changed. The Lewis structure skeleton must not change under any circumstances. All the canonical structures of the required molecule must have an equal number of electrons. Resonance structures should not have a different number of electrons. It can also be defined as the difference between the expected enthalpy and calculated enthalpy.Īs less energy is required to convert a conjugated benzene structure to a stable benzene ring, Benzene has a lower heat of formation. The amount of energy required to convert the conjugated structure or delocalized structure into a stable structure is called a resonance energy. Resonance allows the electrons to relocate from their original positions by lowering their energy to achieve a more stable canonical structure. The canonical structure or resonance structure is a method of embodying the delocalized electrons within the molecule. The above-mentioned structure is the canonical structure of Benzene. This alternate arrangement of double and single bonds in a molecule is termed conjugation. The benzene molecule is different from Cyclohexane as the carbon atoms in the benzene ring are attached by alternate single and double bonds. Let’s Take an Example of a Benzene molecule:. Resonance Hybrid is a combination of all the resonance or canonical structures of that molecule. Resonance represents a particular molecule by comparing two or more Lewis structures where a single Lewis skeleton of that particular compound fails to explain the bonding in a molecule. Resonance structure can also be termed as an intermediate structure between two or more than two structures. The electrons delocalize to make the molecule stable.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
